Modicus Prime Raises $8M to Enable AI Audit Readiness Across Pharma
PR Newswire
AUSTIN, Texas, May 6, 2026
AUSTIN, Texas, May 6, 2026 /PRNewswire/ — Modicus Prime, a former resident at Johnson & Johnson Innovation – JLABS @ Texas Medical Center, announced today the completion of an additional $4.5M in funding led by Frist Cressey Ventures, bringing total funding to $8M with additional backing from Silverton Partners and Oncology Ventures. The funding comes following the multiple rollouts of the Trustworthy AI Compliance Software enabling scalable, audit-ready AI systems across global pharmaceutical companies, CROs, CMOs, CDMOs, and adjacent regulated sectors.
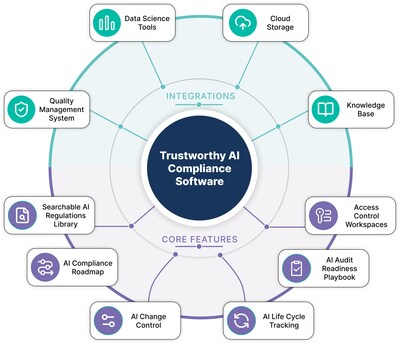
The Trustworthy AI Compliance Software enables pharmaceutical companies’ AI applications in regulated (GxP) environments, both internal and from AI suppliers, to be audit-ready across their organizations. It integrates with pharmaceutical tech stacks and quality management systems, acting as a central layer that enables scalable AI use across sites and geographies with varying regulatory requirements. The integration enables a single source of truth for compliance with global regulators including the FDA and EMA across the full AI lifecycle (see Figure 1). Translating between data science, IT, quality, and compliance, the software reduces operational friction and ensures every pharmaceutical stakeholder works compliantly with AI in regulated environments. The result is safer AI adoption, reduced compliance risk and protected ROI of AI investments, allowing pharma organizations to stay focused on delivering medicines to patients. Compliant AI ultimately accelerates adoption at scale and helps reduce, for example, costly quality control failures costing the industry an estimated $50 billion annually.
FCV founding partner Senator Bill Frist, MD states, “At the end of the day, every advancement in this industry must serve the patient. By simplifying and strengthening AI compliance, Modicus Prime enables pharmaceutical leaders to stay focused on accelerating innovation and delivering life-saving therapies to those who need them most.”
Modicus Prime’s leadership is helping drive the industry’s AI movement through authoring guidelines, keynotes, webinars, and talks informing regulators. Taylor Chartier, Modicus Prime’s CEO, co-authored the ISPE GAMP AI Guide and BioPhorum AI Implementation publication in 2025. She also spearheaded ISPE’s GxP AI Webinar Series with fellow industry experts, which was an industry-first webinar series on implementing AI/ML solutions in GxP environments. Chartier continues to contribute to educational initiatives to upskill the pharmaceutical workforce in partnership with quality-focused organizations, including Pathway for Patient Health.
A former engineer and data scientist from Shire and Bayer Pharmaceuticals, Chartier states, “Pharmaceutical companies are investing heavily in AI, but without the infrastructure to deploy it compliantly and measure ROI, that investment is at risk. Adjacent healthcare sectors, including payers and providers, express the same need for compliance infrastructure. This is the gap our Trustworthy AI Compliance Software addresses, enabling scalable AI adoption across regulated environments.”
Oncology Ventures’ Ben Freeberg states, “The next wave of cancer breakthroughs are coming, and every manufacturer has to survive a regulatory audit to ensure those drugs get to patients who need them. Modicus Prime is the compliance layer that makes this happen.”
Modicus Prime is using this new capital to further service their international pharmaceutical customers with additional customer support, including the continued enhancement of its platform to better support customer deployments and growing AI compliance needs.
About Modicus Prime
Modicus Prime’s AI Compliance platform bridges data science, IT, and quality, enabling regulated organizations to deploy and scale trustworthy AI. By providing a single source of truth for regulators, the platform ensures AI applications remain compliant across their full lifecycle, from development through evolving GxP use. A resident at Johnson & Johnson Innovation – JLABS @ South San Francisco, Modicus Prime is venture-backed and partners with leading pharmaceutical organizations to enable AI audit readiness at scale. The platform integrates with existing tech stacks and quality management systems, supporting biologics, cell and gene therapies, and vaccine manufacturing across global regulatory environments.
About Frist Cressey Partners
Founded by former U.S. Senate Majority Leader Bill Frist, MD and Bryan Cressey, Frist Cressey Ventures (FCV) is a healthcare venture capital firm with $846M AUM focused on accelerating the growth of high potential healthcare companies through a differentiated healthcare network, hands-on leadership support, and healthcare policy expertise. Our mission is simple: to transform healthcare to improve lives. Our strategic partners, including The Cigna Group, MedStar Health and OhioHealth, provide healthcare to more than 50% of the U.S. population. We invest in early-stage startups that build technology or tech-enabled services to advance quality of care, system integration, patient outcomes, affordability, and access. To learn more about Frist Cressey Ventures, visit our website at www.fcventures.com.
About Oncology Ventures
Oncology Ventures is a specialized, Austin-based venture capital firm founded in 2022 by cancer survivor Ben Freeberg. The Fund invests in commercially-validated start-ups transforming the future of cancer care. Oncology Ventures is partnered with leading oncology institutions, including Texas Oncology, Fred Hutch Cancer Center, City of Hope, Moffitt and others.
Contact:
Taylor Chartier
taylor@modicusprime.com
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/modicus-prime-raises-8m-to-enable-ai-audit-readiness-across-pharma-302763769.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/modicus-prime-raises-8m-to-enable-ai-audit-readiness-across-pharma-302763769.html
SOURCE Modicus Prime